T
H E O R Y
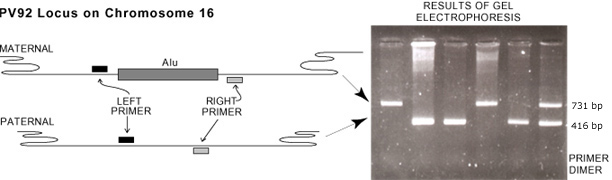
This experiment examines PV92, a human-specific Alu insertion on chromosome 16. The PV92 genetic system has only two alleles indicating the presence (+) or absence (-) of the Alu transposable element on each of the paired chromosomes. This results in three PV92 genotypes (++, +-, or --). The + and - alleles can be separated by size using gel electrophoresis.
Alu elements are classified as SINEs, or Short INterspersed Elements. All Alus are approximately 300 bp in length and derive their name from a single recognition site for the restriction enzyme AluI located near the middle of the Alu element. Human chromosomes contain about 1,000,000 Alu copies, which equal 10% of the total genome. Alu elements probably arose from a gene that encodes the RNA component of the signal recognition particle, which labels proteins for export from the cell.
Alu is an example of a so-called "jumping gene" – a transposable DNA sequence that "reproduces" by copying itself and inserting into new chromosome locations. Alu is classified as a retroposon, because it is thought to require the retrovirus enzyme reverse transcriptase (rt) enzyme to make a mobile copy of itself. Here is a simple scheme to explain how an Alu element transposes:
First, the inserted Alu is transcribed into messenger RNA by the cellular RNA polymerase.
Then, the mRNA is converted to a double-stranded DNA molecule by reverse transcriptase.
Finally, the DNA copy of Alu is integrated into a new chromosomal locus at the site of a single- or double-stranded break.
Each Alu element has an internal promoter for RNA polymerase III needed to independently initiate transcription of itself. However, Alu is a "defective" transposon, in that it lacks the enzyme functions to produce a DNA copy of itself and to integrate into a new chromosome position. However, Alu can obtain these functions from another transposon, called L1, a Long INterspersed Element (LINE). LINEs are essentially defective retroviruses that retain a functional rt gene. Interestingly, in addition to reverse transcribing RNA to DNA, the L1 rt also produces single-stranded nicks in DNA. In the current model, the rt enzyme produces a nick at a chromosomal locus containing the sequence AATTTT. The polyadenylated "tail" of the Alu transcript (-AAAA) then hydrogen bonds to the TTTT sequence at the nick site, creating a primer for reverse transcription. The L1 rt makes a staggered nick in the opposite DNA strand of the host chromosome, allowing the DNA copy to integrate. This method of insertion also accounts for the identical sequences (direct repeats) found at the ends of all Alu elements. So it appears that LI can provide the necessary functions for Alu transposition. In this sense, Alu is a parasite of L1, which, in turn, is a relic of a retrovirus ancestor.
Some scientists regard Alu as an example of "selfish DNA" – it encodes no protein and appears to exist only for its own replication. If one reduces the definition of life to "the perpetuation and amplification of a DNA sequence through time," then Alu is an extremely successful life form. However, other scientists believe that transposable elements have played an important role in evolution by creating new mutations and gene combinations. Nobel laureate Barbara McClintock hypothesized that transposable elements provide a mechanism to rapidly reorganize the genome in response to environmental stress. Like Alu, the Ds transposable element discovered in corn by McClintock is a defective transposon and requires the help of a second element called Ac (activator).
Alu elements are found only in primates – the "monkey" branch of the evolutionary tree, which includes humans. So, all of the hundreds of thousands of Alu copies have accumulated in primates since their separation from other vertebrate groups about 65 million years ago. Once an Alu integrates into a new site, it accumulates new mutations at the same rate as surrounding DNA loci. Alu elements can be sorted into distinct lineages, or families, according to inherited patterns of new mutations. These studies suggest that the rate of Alu transposition has changed over time – from about one new jump in every live birth, early in primate evolution, to about one in every 200 newborns today. Taken together, this pattern suggests that, at any point in time, only one or several Alu "masters" are capable of transposing.
Once an Alu inserts at a chromosome locus, it can copy itself for transposition, but there is no evidence that it is ever excised or lost from a chromosome locus. So, each Alu insertion is stable through evolutionary time. Each is the "fossil" of a unique transposition event that occurred only once in primate evolution. Like genes, Alu insertions are inherited in a Mendelian fashion from parents to children. Thus, all primates showing an Alu insertion at a particular locus have inherited it from a common ancestor. This is called identity by descent.
An estimated 500-2,000 Alu elements are restricted to the human genome. The vast majority of Alu insertions occur in non-coding regions and are thought to be evolutionarily neutral. However, an Alu insertion in the NF-1 gene is responsible for neurofibromatosis I, Alu insertions in introns of genes for tissue plasminogen activator (TPA) and angiotensin converter enzyme (ACE) are associated with heart disease. Alu insertions are analogous to the insertion of a provirus in viral diseases and certain cancers.
Most Alu
mutations are "fixed," meaning that both of the paired chromosomes have
an insertion at the same locus (position). However, a number of human-specific
Alus are dimorphic – an insertion may be present or absent on each
of the paired chromosomes of different people. These dimorphic Alus
inserted within the last million years, during the evolution and dispersion
of modern humans. These dimorphisms show differences in allele and genotype
frequencies between modern populations and are tools for reconstructing
human prehistory.
Noncommercial, educational use only.